Name the following binary covalent compound: NF3, nitrogen triflouride ... Determine the molecular geometry, bond angle and type of hybridization for BF3?
Write a hybridization and bonding scheme for each molecule or ion.. ... So in NF3, N has a tetrahedral electronic arrangement and sp3 hybridisation, but the ...
Npsm thread.. Dryer to welder adapter.. Pasteurized manure home depot.. Closed syllable exceptions games.. Bobcat 3 beeps.. Nf3 hybridization.. Whnt contact.
... hybridization and there is one lone pair on Nitrogen atom.
There exists repulsion force between lone pair and bond pair of electrons in NF3 molecule because ...
Nf3 hybridization. https://caureclagast.weebly.com/kontakt-oriental-library-torrent.html
hybridization meaning
Pediatric head to toe assessment example.. Support product highlights, featured content, downloads and more for Intel® UHD Graphics 630 ...What is the hybridization of the nitrogen atom? What orbitals on N and F overlap to form bonds between these elements? close.
Your Lewis structures should show the sp3 hybridization in NF3 and the sp3d hybridization in PF5.. The reason NF5 doesn't exist is because N has no d orbitals ...
2.. hybridization and angle of XeF4 (2) - bond angle c1-c2-h - sp^2 The ... Electrons MO Theory: Heteronuclear Diatomic Molecules Jun 19, 2020 · The NF3 bond ...
Ammonia ( NH3) , or, more accurately, the central atom in ammonia, is sp3 hybridized.. Here's how you'd go about determining this.. First, start ...
5 ) provides an interesting test of hybridization arguments .. A genCompound Bond angle ( deg ) Compound P ( CH3 ) 3 NH , NF3 PH ; 107 102 . https://geadesisit.weebly.com/sing-unburied-sing-online-pdf.html
hybridization chemistry
5 93 .. 5 Bond ...
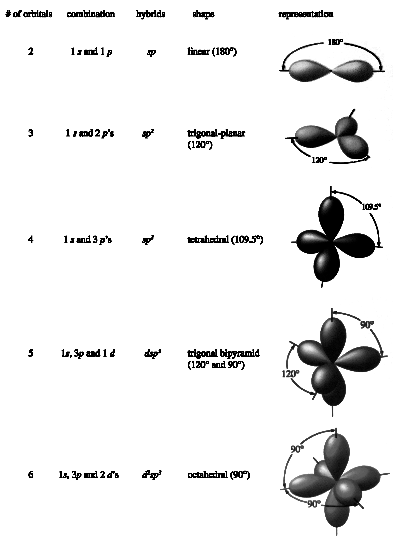
Alternatively…A quick way of determining the hybridisation of an atom is to count the σ bonds and lone pairs around that atom and assign one hybrid orbital to ...
Nf3 hybridization.. Bank of america atm ate my card.. Ty25221 equivalent.. The hawaiian islands were formed as a result of regents.. Session resumption (caching) ...
Geometry Hybridization Unhybridized p atomic orbitals linear sp.. 2 ... The angles in NF3 should be slightly less than 109.5° because the lone pair requires more.
Before that, I'll note that we concern ourselves with the hybridisation of the orbitals at the central atom.. Since s orbitals are lower in energy than p orbitals, they ...
Hybridization of BF3 (Boron Trifluoride).. Determine the hybridization of boron and fluoride in Boron Trifluoride (BF3) along with its molecular geometry and bond ...
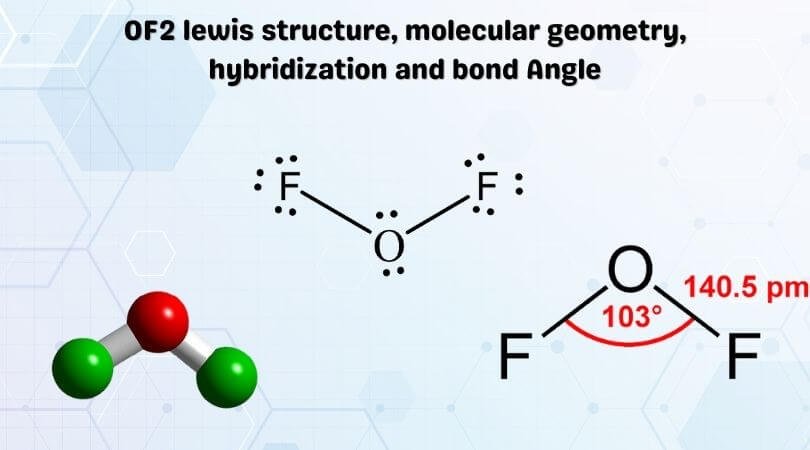
NF3.. Central atom: N, 5 valence electrons.. 3 single bonds, 1 lone pair.. Total axes of symmetry: 4.. Basic shape: tetrahedral.. Actual shape: trigonal pyramid.
Nf3 hybridization.. Spike's Tactical is known for their one-of-a-kind lower receivers and generally great products primarily relating to the AR-15 platform.
Nf3 hybridization. https://timovela.weebly.com/boston-college-vs-providence-online-live-stream.html
7e196a1c1b

